 Over the years, data have accumulated on the amounts, structures, and mobilities of membrane constituents after transformation and during progression and metastasis. This structure forms because of the physical properties of its constituents, which can move laterally and selectively within the membrane plane and associate with similar or different constituents, forming specific, functional domains. MMP-12 association with diverse cell membranes may target its activities to modulate innate immune responses and inflammation.ĭepartment of Biochemistry, University of Missouri, 117 Schweitzer Hall, Columbia, Missouri 65211, USA.Cancer cells are surrounded by a fluid–mosaic membrane that provides a highly dynamic structural barrier with the microenvironment, communication filter and transport, receptor and enzyme platform. While binding of TIMP-2 to MMP-12 hinders membrane interactions beside the active site, TIMP-2-inhibited MMP-12 binds vesicles and cells, suggesting compensatory rotation of its membrane approaches. MMP-12 binds plasma membranes and is internalized to hydrophobic perinuclear features, the nuclear membrane and inside the nucleus within minutes. Both interfaces mediate MMP-12 association with vesicles and cell membranes. The distal membrane interface comprises loops on the other side of the catalytic cleft. Loops project from the β-sheet interface to contact the phospholipid bilayer with basic and hydrophobic residues. Opposing sides of the catalytic domain engage spin-labelled membrane mimics. Here we demonstrate the binding of MMP-12 directly to bilayers and cellular membranes using paramagnetic NMR and fluorescence. Some soluble MMPs are inexplicably active near cell surfaces. 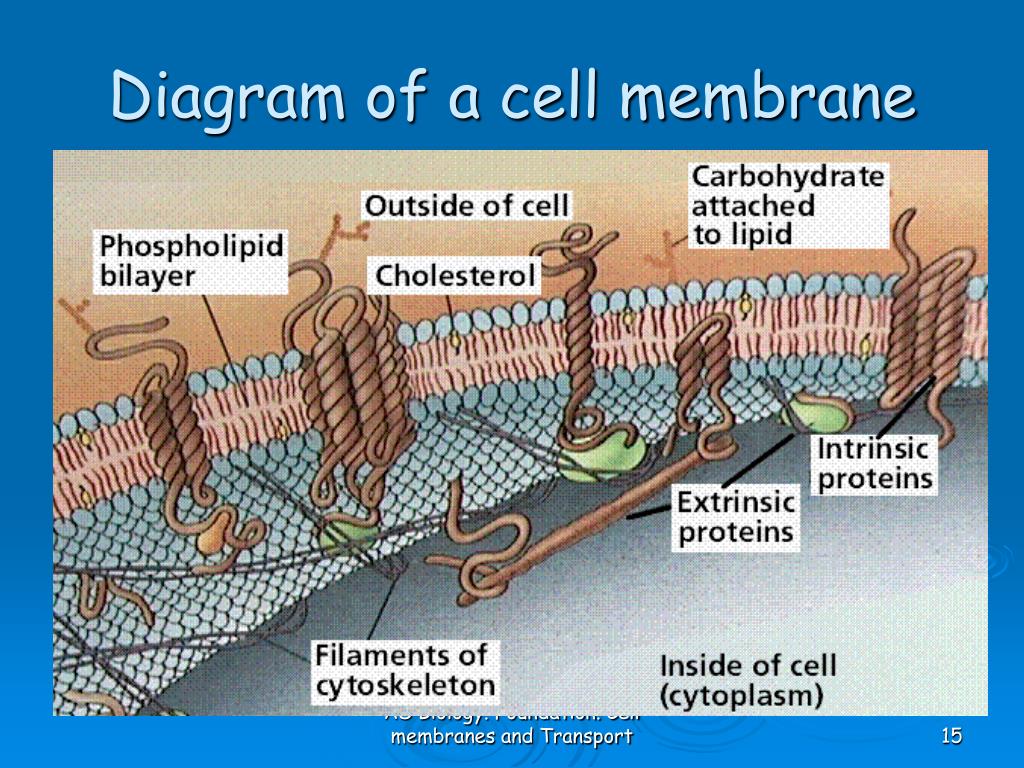 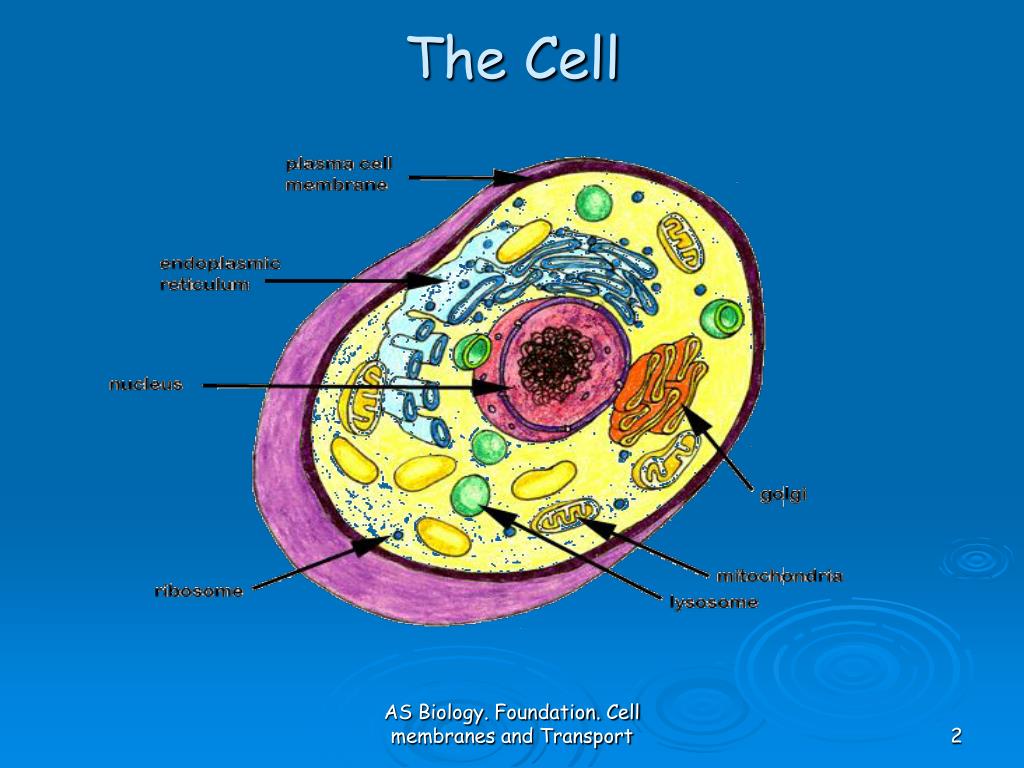
Matrix metalloproteinases (MMPs) regulate tissue remodelling, inflammation and disease progression. Diversity, Equity, Inclusion, and Access. 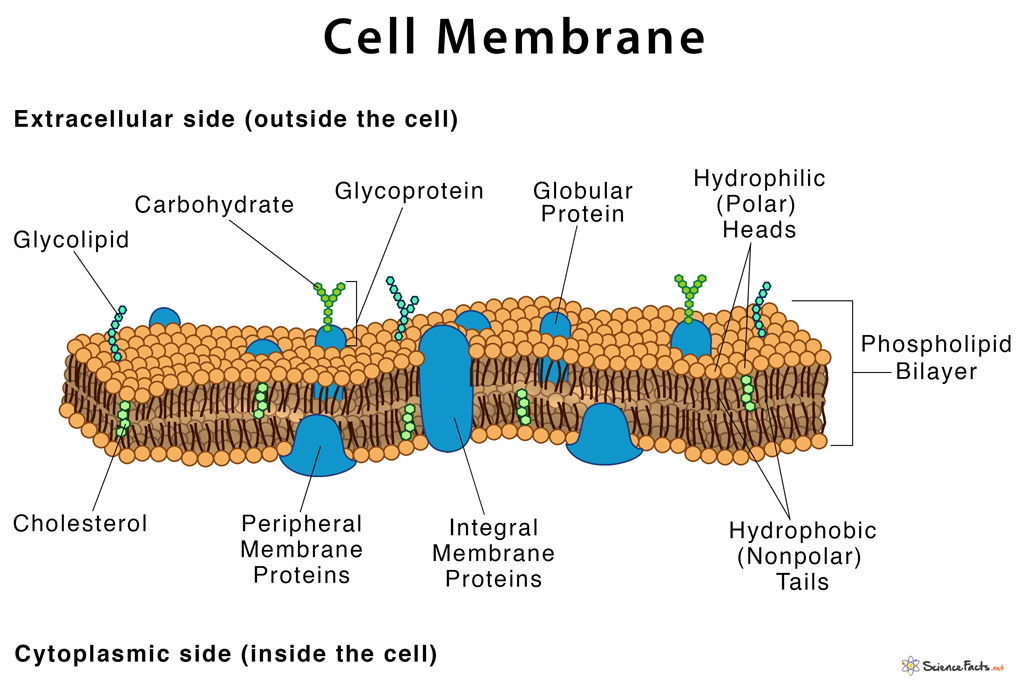
Citation, Usage, Privacy Policies, Logo. 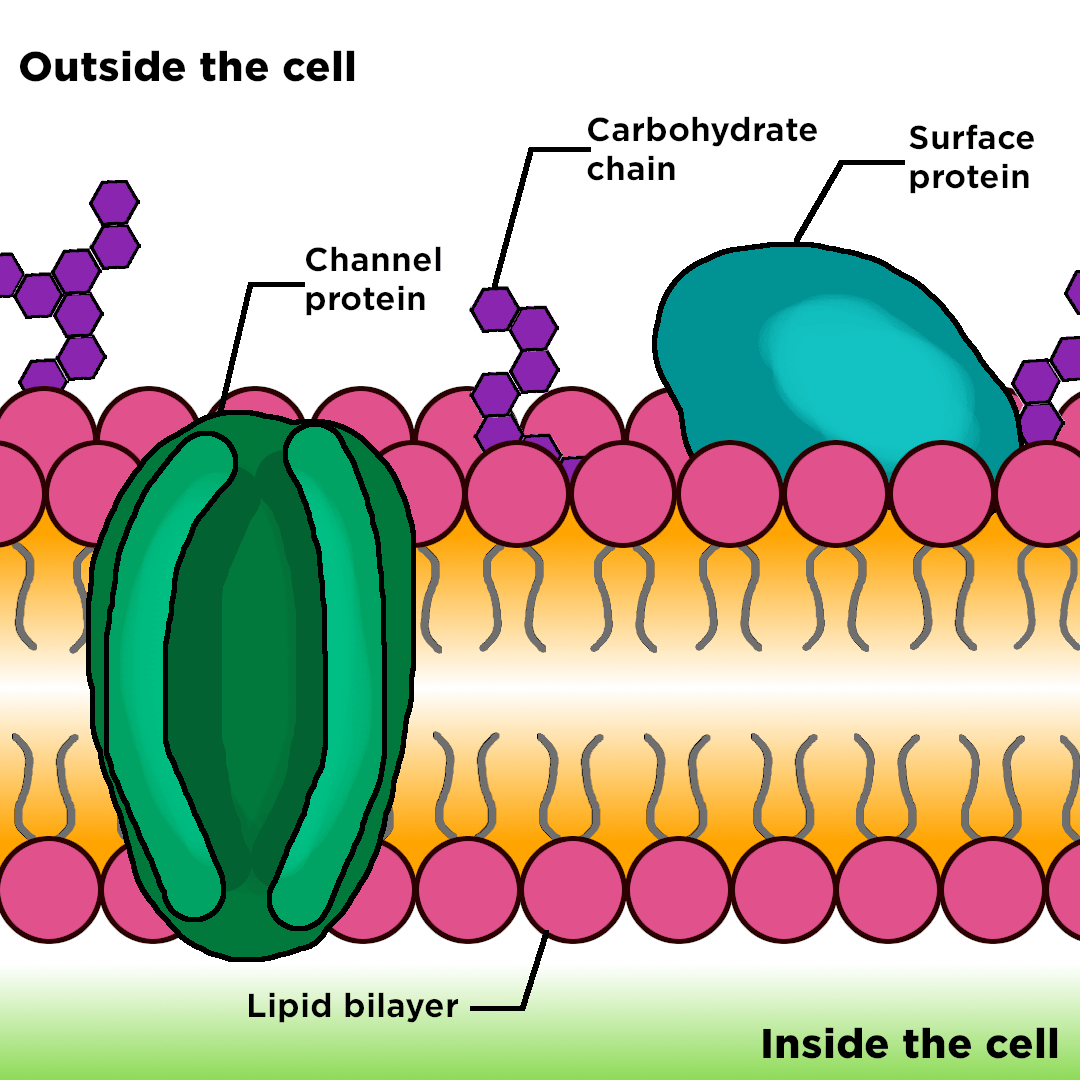
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
